Vein Services
Understanding Superficial Venous Reflux
Your legs are made up of a network of veins. Healthy leg veins contain valves that open and close to assist the return of blood back to the heart. Venous reflux disease develops when the valves that keep blood flowing out of the legs and back to the heart become damaged or diseased. This can cause blood to pool in your legs and lead to symptoms such as pain, swelling, swollen limbs, leg heaviness and fatigue, skin changes and skin ulcers, and varicose veins.

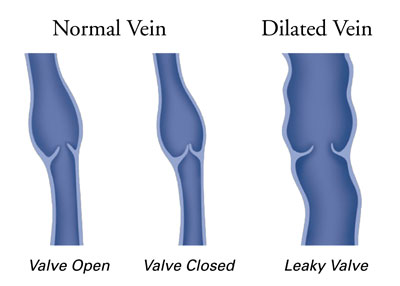
Introducing VNUS
At Biltmore Cardiology we are pleased to provide the VNUS Closure® procedure, in addition to other treatment modalities for comprehensive management of vein disease. The Closure procedure is a minimally invasive treatment alternative for patients with a condition known as superficial venous reflux, which often results in varicose veins and other painful symptoms. Varicose veins are known for their distinctive blue color and bulging appearance under the skin. They afflict an estimated 10%-20% of the adult population. In the United States, this translates into 20-25 million people, the majority of whom are women.
The Closure® Procedure
The VNUS® Closure procedure, an alternative treatment option to traditional vein stripping surgery, brings state-of-the-art technology to an age-old disease.
The Closure procedure is performed on an outpatient basis. Using ultrasound, your physician will position the Closure catheter into the diseased vein, through a small opening in the skin. The tiny catheter delivers radiofrequency (RF) energy to the vein wall. As the RF energy is delivered and the catheter is withdrawn, the vein wall is heated, causing the collagen in the wall to shrink and the vein to close. Once the diseased vein is closed, blood is re-routed to other healthy veins.
Following the procedure, a simple bandage is placed over the insertion site, and additional compression may be provided to aid healing. Your doctor may encourage you to walk, and to refrain from extended standing and strenuous activities for a period of time.
Patients who undergo the Closure procedure typically resume normal activities within a day.
Is Closure Right for Me?
As with any medical procedure, the Closure¨ procedure is not suitable for all people and there are associated risks and considerations. You should consult your physician to determine if the Closure procedure is right for you, and to discuss any special risks your individual condition may present.
Indications:
The Closure System is intended for endovascular coagulation of blood vessels in patients with superficial venous reflux.
Contraindications:
Patients with thrombus in the vein segment to be treated.
PRECAUTION: For patients with a pacemaker, internal defibrillator or other active implanted device, consult the cardiologist and the manufacturer of the active implanted device. Continuous patient monitoring during the procedure is recommended. Evaluate the patient and the implanted active device post procedure. Keep all power cords and the attached sterile cable away from the location of the pacemaker or leads, defibrillator or the implanted active device.
CAUTION: In patients with an aneurysm in the vein segment to be treated, the vein wall may be thinner in the area of the aneurysm. To effectively occlude a vein with an aneurysmal segment, additional tumescent infiltration may be needed over the aneurysmal segment, and the treatment of the vein should include segments proximal and distal to the aneurysmal segment.
CAUTION: No data exists regarding the use of this catheter in patients with documented peripheral arterial disease. The same care should be taken in the treatment of patients with significant peripheral arterial disease as would be taken with a traditional vein ligation and stripping procedure.
Potential Risks & Complications
- Potential complications include, but are not limited to, the following: vessel perforation, thrombosis, pulmonary embolism, phlebitis, hematoma, infection, adjacent nerve injury, skin burns.
- Treatment of veins located very close to the skin surface may result in a skin burn.
- Adjacent nerve injury is higher with treatment at or below the calf.
VNUS Clinical Outcomes
Clinical results indicate that the Closure procedure can be effective at closing the vein and significantly reducing patient symptoms.
(Note: These results were obtained using VNUS Closure and VNUS ClosurePlus; RF products with expandable extrodes)
At two years following treatment, patients who had undergone the Closure procedure reported superior quality of life compared to vein stripping patients.1
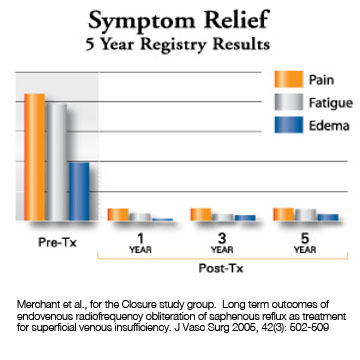
The Closure procedure has proven to be highly effective, with 87% of veins treated remaining closed at five years. Results also demonstrate long-term relief of symptoms.2
Patient Symptoms Reported at Follow-up
Three randomized trials of the Closure procedure versus vein stripping, including the multi-center comparative trial published August 2003 in the Journal of Vascular Surgery, show very similar results.2,3,4 In the most recent trial every statistically significant outcome was in favor of the Closure procedure, resulting in1:
- Experienced less post-operative pain and bruising
- Resumed normal activities within one day
- Returned to work more than a week earlier than vein stripping patients
Visual Results

References
1 Lurie F, Creton D, Eklof B, Kabnick LS, Kistner RL, Pichot O, et al. Prospective randomized study of endovenous radiofrequency obliteration (Closure) versus ligation and stripping in a selected patient population (EVOLVES study). J Vasc Surg 2003;38:207-14.
2 Merchant RF, et al. J Vasc Surg 2005; 42: 502-9
3 Stštter L. Schaaf I, Fendl R, Bockelbrink A. Randomized Study to Compare the Closure Procedure, Invagination Stripping and Cryo Stripping for Treatment of the Greater Saphenous Vein. Abstract submitted to German Society for Phlebology for presentation Sept. 2003.
4 Rautio T, Ohinmaa A, PerŠlŠ J, Ohtonen P, Heikkinen T, Wiik H, et al. Endovenous obliteration versus conventional stripping operation in the treatment of primary varicose veins: A randomized controlled trial with comparison of costs. J Vasc Surg 2002;35:958-65.
